About the Bavarian Cancer Registry
The Bavarian Cancer Registry
Structure
The legal basis of the cancer registration in Bavaria is constituted by the State Law on the Bavarian Cancer Registry (Bayerisches Krebsregistergesetz, BayKRegG).
Coordination Office
The Coordination Office is responsible for the central definition of standard operation procedures with the aim of a statewide harmonisation of methods and quality assessment. The Coordination Office supports the cancer registry's advisory board.
Center of Early Cancer Detection and Cancer Registration
The Center of Cancer Registration (Registry Office) collects data from the Regional Registration Centers and is responsible for statewide clinical-epidemiological analyses. Data and findings are published in annual reports. The epidemiological data are supplied once a year to the Central Cancer Surveillance Programme at the Robert Koch Institute Berlin, which provides information regarding the epidemiological data on cancer nationwide in Germany.
Anonymous data can be provided for research purposes.
Regional Registration Centers
The former clinical registries located in Augsburg, Bayreuth, Erlangen, München, Regensburg and Würzburg now act as Regional Cancer Registry Centers of the Bavarian Cancer Registry. They collect all cancer notifications from their region, stay in contact with their reporters and project groups, and keep them up-to-date with feedback and regional analyses.
Trust Center
The Confidentiality Office or Trust Center handles patient identity data and is separate from other parts of the cancer registry. The Trust Center is responsible for the registration of death certificates, for health insurance accounting, pseudonymisation of identity data and handling of patients´ opt-out requests.
Organisation
Organisation chart (German only)
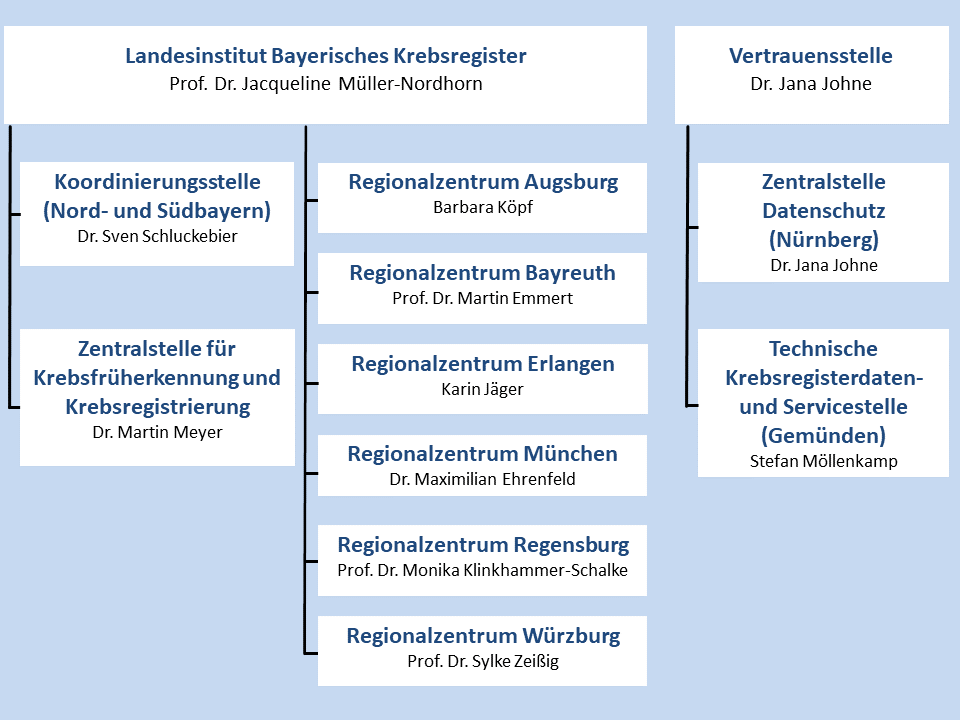
Head: Prof. Dr. med. Jacqueline Müller-Nordhorn, DPH
Contact: Jehona Vatovci, Team assistance
Tel.: +49 (0)9131 6808-4020
E-Mail: Jehona.Vatovci@lgl.bayern.de
Head of Subdivisions
Central
Coordination Office: Dr. med. Sven Schluckebier
Center of Early Cancer Detection and Cancer Registration: Dr. rer. biol. hum. Martin Meyer
Regional Registration Centers
Augsburg: Dr. Stephanie Biber
Bayreuth: Prof. Dr. Martin Emmert
Erlangen: Karin Jäger
München: Dr. Maximilian Ehrenfeld
Regensburg: Prof. Dr. Elisabeth Sturm-Inwald
Würzburg: Prof. Dr. med. Sylke Zeißig
Trust Center
Dr. Jana Johne
Advisory Board
The advisory board of the Bavarian Cancer Registry is convened by the Bavarian State Ministry of Health and Care according to the law (Art. 14 BayKRegG). It supports the Bavarian Health and Food Safety Authority (Bayerisches Landesamt für Gesundheit und Lebensmittelsicherheit, LGL). Furthermore, it coordinates the scientific use of cancer registry data and appraises external data requests.
The advisory board meets at least once a year. The board consists of 18 members from different institutions and fields of work. The State Ministry chairs the advisory board.
Partner
Platform § 65c
The Platform § 65c is the association of all clinical cancer registries in Germany in accordance to the federal cancer registration law.
The committee of experts coordinates technical issues and procedures between the cancer registries in the federal states and develops and approves recommendations.
The committee provides information about its work on its own website:
Platform § 65c

German Cancer Registries (GNCR)
The Bavarian Cancer Registry is a founding member of the Association of Epidemiological Cancer Registries in Germany (GEKID) - now 'Deutsche Krebsregister e.V. (DKR)'. The main task of the association is to achieve nationwide methodical uniformity by using national and international standards, in spite of different laws regarding cancer registration in the federal states. Moreover, DKR serves as central partner for the cancer registries in cross-state matters.
International Collaboration
The Bavarian Cancer Registry is a member of the European Network of Cancer Registries (ENCR) and of International Association of Cancer Registries (IACR).
Robert Koch Institute
Annually the Bavarian Cancer Registry supplies the epidemiological data to the Central Cancer Surveillance Programme at the Robert Koch Institute in Berlin, where the epidemiological data of all federal states are gathered and evaluated nationwide. The completeness of the cancer registration is estimated. The report "Cancer in Germany" (Krebs in Deutschland) is based on the data collected by the federal states.
Recorded Data Set
The reported data items are determined by law. They include the German-wide joint dataset defined by the Association of German Tumor Centers (Arbeitsgemeinschaft Deutscher Tumorzentren, ADT) and DKR („onkologischer Basisdatensatz“). Besides personal identifiers (names and address), the data set comprises information on the incident tumor (diagnosis, location, histology), staging (e.g. TNM), treatment information (curative/palliative surgery, chemo-/radiotherapy) and information about the course of disease.
Documentation is done according to the guidelines of the European Network of Cancer Registries (ENCR) and the Association of Cancer Registries in Germany (DKR).
Data Confidentiality
Data is collected, stored and released according to all legal requirements related to data confidentiality and security. The Trust Center is organised independently from the other parts of the Registry to ensure that the permanently stored medical information cannot be linked to individuals by unauthorized persons.
After pseudonymisation it is not possible to identify individuals anymore. Matching duplicate data records remains possible with pseudonymised data. A decoding of the data is not possible.
The systems used for data storage and processing are strictly secured against unauthorized access. They are separated from the systems supplying publicly available information.
Examples for pseudomized identification items (shortened strings):


