Vector-borne Infectious Diseases in Climate Change Investigations (VICCI):
Projekt 6: Autochthone Leishmaniose in Bayern: Untersuchungen zur Vektorprävalenz und zur Existenz tierischer Reservoirs
Autochthonous leishmaniasis in Bavaria: Investigations of the vector prevalence and of the existence of animal reservoirs
Wissenschaftlicher Abschlussbericht
Projektteilnehmer: Prof. Dr. med. Christian Bogdan, PD Dr. rer. nat. Ulrike Schleicher, Dipl.-Biol. Simone Häberlein, Kirstin Castiglione
Mikrobiologisches Institut – Klinische Mikrobiologie, Immunologie und Hygiene, Friedrich-Alexander-Universität Erlangen und Universitätsklinikum Erlangen
Kontakt:
Prof. Dr. Christian Bogdan
Mikrobiologisches Institut – Klinische Mikrobiologie, Immunologie und Hygiene, Friedrich-Alexander-Universität Erlangen und Universitätsklinikum Erlangen, Wasserturmstraße 3/5, D-91054 Erlangen; christian.bogdan@uk-erlangen.de, Tel. 09131-852-2551
Abstract
Zoonotic leishmaniasis is caused by protozoan parasites of the genus Leishmania, which are transmitted by sand fly vectors from reservoir animals to humans. Within Europe the disease is highly prevalent in Mediterranean countries. In recent years a northward spread of leishmaniasis into previously non-endemic areas of Central Europe was observed. Within Germany, sand flies were already detected in the Federal State of Baden-Württemberg, and four cases of autochthonous leishmaniasis have recently been reported from Bavaria. In the present study, we investigated whether sand flies are prevalent in Bavaria and whether wild rodents carry Leishmania parasites and therefore might function as reservoir animals. In addition, a systematic screening of domestic animals from Bavaria for the prevalence of autochthonous leishmaniasis was performed. For trapping of sand flies, 202 CDC light traps were placed overnight in 155 different locations throughout Bavaria in a series of field trips during the summer months (June to August) of 2009 and 2010. The locations were selected based on different epidemiological and climate parameters that were assumed to favor sand fly occurrence. All light traps were negative for sand flies. Wild rodents were captured in Bavaria and Baden-Württemberg in the years 2001 to 2008. Livers and ear skin were analyzed for the presence of Leishmania DNA by mini exon PCR. The analysis of 300 rodents from six different species (140 Myodes glareolus, 127 Apodemus flavicollis, 17 Apodemus sylvaticus, 14 Microtus arvalis, 1 Mus musculus, 1 Arvicola amphibius) and two specimens of insectivores (Sorex araneus, Sorex coronatus) did not reveal any evidence for an infection with Leishmania parasites. Serum and blood samples from 46 domestic animals (25 horses, 10 dogs, 11 cats) with negative travel history were negative for Leishmania-specific antibodies and viable Leishmania parasites. These results suggest that Bavaria is currently not an established area for the occurrence of sand flies and for autochthonous leishmaniasis in potential reservoir hosts. However, the data do not exclude the possibility of sporadic cases of autochthonous Leishmania infections. Regular surveys and risk analyses need to be performed in the future in order to detect relevant epidemiological changes.
Introduction
The establishment and autochthonous transmission of a vector-borne infectious disease in a new, presently non-endemic region is dependent on the presence of (1) the pathogen itself, (2) reservoir hosts, and (3) a competent vector. Protozoan parasites of the genus Leishmania are transmitted by sand flies and cause cutaneous, mucocutaneous or visceral leishmaniasis in tropical and subtropical countries worldwide. The species Leishmania (L.) infantum is mostly prevalent in Mediterranean countries where it causes visceral leishmaniasis and is mainly transmitted by sand flies of the genus Phlebotomus (Ph.) perniciosus (Killick-Kendrick 1990, Ready 2008). The pathogen is regularly imported into Germany, a presently non-endemic country, by infected travellers and their dogs. Conservative estimates state that about 20,000 dogs infected with Leishmania during a stay in endemic countries are presently living in Germany (Weitzel et al. 2005, Lozán et al. 2008, Menn et al. 2010). Thus, the main limitation to zoonotic leishmaniasis transmission in Germany is the availability of a phlebotomine vector. Since temperature is the most important habitat factor for sand flies, their occurrence in Central Europe is presently restricted to certain climatically favored parts. However, increasing temperatures as a consequence of climate change will likely lead to a spread of sand flies into presently non-endemic regions (Aspock et al. 2008, Fischer et al. 2010). This is already clearly visible in several Central European countries where sand flies were recently discovered, such as Switzerland and Austria (Grimm et al. 1993, Naucke et al. 2011). Also, in Germany sand flies of the species Ph. mascittii and Ph. perniciosus have been frequently detected along the Rhine valley in the Federal State of Baden-Württemberg since 1999 (Naucke and Pesson 2000, Naucke et al. 2008). This indicates a risk for endemic leishmaniasis. Indeed, in Bavaria one case of autochthonous human leishmaniasis and three cases of autochthonous canine and equine leishmaniasis have been reported in the years 1991-2000 (Gothe 1991, Bogdan et al. 2001, Koehler et al. 2002). This ultimately suggests that infected sand flies must be present at least periodically in Bavaria. Thus, a systematic survey for sand flies in this area is an urgent need. Besides domestic animals also wild rodents may play a role as reservoir hosts (Englund and Pringle 2003, Quinnell and Courtenay 2009, Maia et al. 2010). L. infantum infection of the Brown rat (Rattus norvegicus) and Black rat (Rattus rattus) was reported in several Mediterranean countries (Gradoni et al. 1983, Martin-Sanchez et al. 2004, Papadogiannakis et al. 2010, Psaroulaki et al. 2010). In Iran rodents are even described as the main reservoir hosts for zoonotic leishmaniasis caused by L. major (Rassi et al. 2006, Pourmohammadi et al. 2008). Thus, the presence of Leishmania-infected rodents in Bavaria could serve as an indicator for the presence of Leishmania-transmitting sand flies.
Objectives
In this study, for the first time a survey for the prevalence of sand flies and of autochthonous Leishmania-infection in rodents and domestic animals was conducted in Bavaria in order to assess the risk for an establishment of leishmaniasis in this region. In collaboration with Prof. Carl Beierkuhnlein (VICCI project 7: Biogeographical analysis of health-relevant species and projection of their spreading tendencies under changing climatic conditions in Bavaria), focus regions for sand fly trapping in Bavaria were defined that were assumed to be most suitable for sand fly occurrence based on the evaluation of epidemiological and climate parameters. A field survey in these regions was conducted in the years 2009 and 2010 to reveal the vector prevalence. In order to assess the prevalence of autochthonous leishmaniasis in reservoir animals within Bavaria, samples from wild rodents that were trapped in the years 2001-2008 and provided by Dr. Sandra Eßbauer (VICCI project 5: Study on the presence of rodent-transmitted zoonoses along a climate gradient in the Bavarian Forest National Park) as well as serum and blood samples from domestic animals provided by veterinarians in Bavaria were screened for Leishmania DNA and Leishmania-specific antibodies, respectively.
Material and methods
Definition of focus regions for sand fly trapping
Sand fly trapping was performed in focus regions in Bavaria that were defined on the basis of the following epidemiological and climate parameters: (1) locations of autochthonous leishmaniasis cases in Bavaria that indicate a sporadic occurrence of Leishmania-infected sand flies (Gothe 1991, Bogdan et al. 2001, Koehler et al. 2002); (2) evidence for sand flies in neighboring states which would favor sand fly immigration into Bavaria (Grimm et al. 1993, Naucke et al. 2008); (3) present climate conditions in Bavaria based on data from the German meteorological service (Naucke et al. 2008), since temperature is a main habitat factor for sand fly survival (Cross and Hyams 1996, Aspock et al. 2008, Fischer et al. 2010); (4) temperature-derived risk maps for the sand fly distribution in Germany for the time period 2011-2040 (Fischer et al. 2010). A focus region was defined as such when at least two of the four epidemiological and climate parameters were fulfilled.
Trapping of sand flies
The entomological survey in the previously defined focus regions of Bavaria was carried out in the years 2009 and 2010 in the warm season from end of June until mid of August, when the climatic conditions in Germany are thought to favor sand fly activity (Naucke et al. 2008). Presence of phlebotomine vectors was assessed by Centres for Disease Control and Prevention (CDC) New Standard Miniature Light Traps (model 1012; John W. Hock Company, Gainesville, Florida, USA). Traps were fixed 1 m above the ground and were preferentially set close to human dwellings, especially on farms close to animal shelters or in old barns with non-concreted substrate, humid and organic cover because of the reported habitat preference of sand flies (Grimm et al. 1993, Naucke and Pesson 2000). Traps were run overnight, for one night or two consecutive nights, and collected the next morning. Night temperature, wind and rain conditions, GPS coordinates, as well as characteristics of the trap location (e.g. material of the building and substrate) and presence of domestic animals were recorded. Night temperatures were mostly above 12 °C. Captured insects were stored in 70% ethanol or frozen at ?20 °C for later morphological identification by microscopic analysis.
Trapping of rodents
Rodents were collected by co-workers of Dr. Sandra Eßbauer (Bundeswehr Institute of Microbiology, Munich, Germany) from 2001-2008 using Sherman live traps at different locations in southern Germany within the following administrative districts (months of capture): (a) Regen (October 2004, May 2005, September-October 2008), (b) Freyung-Grafenau (October 2004, May-September 2005, September-October 2008), (c) Freising (October 2001), (d) Fürstenfeldbruck (November 2002, January and April 2003), (e) Munich (October-November 2001 and 2002), and (f) Traunstein (May and July 2003) in the Federal State of Bavaria; (g) Sigmaringen (July-August 2007) and (h) Konstanz (September 2002) in the Federal State of Baden-Württemberg. Some rodents were trapped in other districts of Bavaria (Altötting in October 2002, Erding in February 2003). The capture sites included forests, fields, grasslands, blackberry bushes and farms. Animals were trapped after decoying using 0.5 liter plastic cups with apple pieces as bait. Traps were controlled twice a day. Animals were anesthetized by CO2 exposure and killed humanely according to the German Law of Animal Protection.
Preparation of rodent tissue samples and DNA isolation
Tissue samples (liver and ears) from rodents were aseptically removed and homogenized. Samples from mice trapped in the administrative districts Feyung-Grafenau, Regen and Sigmaringen were homogenized in minimal essential medium supplemented with 3% fetal calf serum (Biochrom AG, Berlin, Germany) using Lysing matrix tubes A (MP Biomedicals, Eschwege, Germany) and a Fast Prep 120 instrument (Qbiogene, Heidelberg, Germany) by co-workers of Dr. Sandra Eßbauer. Nucleic acids were prepared from these tissue homogenates using the QIAamp Viral RNA Mini Kit (Qiagen, Hilden, Germany). This test was used because of investigations on RNA-viruses were conducted in parallel and it yields a quality of extracted DNA comparable with other standard DNA extraction kits (Schex et al. 2011). For DNA isolation from all other rodent samples, different buffers for tissue homogenization using a TissueLyser (Qiagen) and different methods for DNA isolation were tested: (a) tissue homogenization in PBS, DNA isolation with QIAamp Viral RNA Mini Kit; (b) tissue homogenization in Trizol (Invitrogen, Darmstadt, Germany), DNA isolation with chloroform and ethanol treatment; (c) tissue homogenization in RLT Plus buffer (Qiagen), DNA isolation with AllPrep DNA/RNA Kit (Qiagen). The extracted DNA was stored at ?20 °C.
PCR analysis of rodent DNA
DNA from livers and ear skin of rodents was analyzed for the presence of Leishmania DNA using a mini exon PCR. The sensitivity of the PCR was optimized on pure Leishmania DNA prior to use for diagnosis. PCR amplification was carried out in a GeneAmp PCR System 9700 (Applied Biosystems, Darmstadt, Germany). For mini exon PCR, the primers Fme (5?-TAT TGG TAT GCG AAA CTT CCG-3?) and Rme (5?-ACA GAA ACT GAT ACT TAT ATA GCG-3?) (Thermo Scientific, Braunschweig, Germany) were used to amplify the nontranscribed spacer region of the mini exon sequence of Leishmania (Marfurt et al. 2003). After an initial denaturation step (5 min at 94 °C), 35 cycles of 94 °C for 30 s, 50 °C for 30 s (annealing), and 72 °C for 45 s (extension) were performed, followed by 10 min at 72 °C. As a positive control genomic DNA of L. major, L. infantum and L. tropica promastigotes was used. To proof the quality of DNA preparation, all PCR samples were tested for the murine house-keeping gene ?-actin using the primers p1 (5?-CAC CCG CCA CCA GTT CGC CA-3?) and p2 (5?-CAG GTC CCG GCC AGC CAG GT-3?) (Thermo Scientific) and the following PCR conditions: 5 min at 94 °C, 35 cycles of 95 °C for 30 s, 58 °C for 30 s and 72 °C for 45 s, and a final 10 min at 72 °C. Amplicons were analyzed on 2.5% (mini exon) or 1.5% (?-actin) agarose gels by electrophoresis at 120 V in 1× Tris-acetate-EDTA buffer.
For initial establishment of a screening method for Leishmania DNA, some rodent DNA samples were screened for Leishmania SSU rDNA by using real-time RT-PCR (Wortmann et al. 2001), or by conventional PCR with primers for kinetoplast DNA (Rodgers et al. 1990) or 18S rDNA (Schulz et al. 2003). For kinetoplast PCR the primers Lei 13A (5´-GTG GGG GAG GGG CGT TCT-3´) und Lei 13B (5´-ATT TTA CAC CAA CCC CCA GTT-3´) were used with the following cycle conditions: 3 min at 94 °C, 40 cycles of 94 °C for 30 s, 55 °C for 30 s and 72 °C for 30 s, and a final 5 min at 72 °C. The 18S rDNA primers CDLS (5?-GCT CCA AAA GCG TAT ATT AAT GCT GT-3?) and CDLA (5?-TCC TTC ATT CCT AGA GGC CGT GAG T-3?) were applied in a conventional PCR with 10 min at 94 °C, 35 cycles of 94 °C for 60 s, 55 °C for 60 s and 72 °C for 60 s, and a final 10 min at 72 °C. For detection of Leishmania SSU rDNA by real-time RT-PCR the Leishmania-specific primer LEIS.U1 (5´-AAG TGC TTT CCC ATC GCA ACT-3´), the reverse primer LEIS.L1 (5´-GAC GCA CTA AAC CCC TCC AA-3´), and the Leishmania-specific probe LEIS.P2 (fam-TTC GGT GTG TGG CGC C-nfq-mgb) were used. PCR amplification and detection were performed with the real-time PCR system ABI Prism 7900 HT (Applied Biosystems, Darmstadt, Germany). The cycle conditions used were 50 °C for 2 minutes and 95°C for 10 min, followed by 45 cycles of 2-step incubations at 95°C for 15 seconds and 60°C for 60 seconds.
Blood samples from domestic animals
Veterinarians from Bavaria provided serum and EDTA blood samples obtained from horses, dogs and cats in the years 2010 and 2011. Serum was screened by Western blot and immuno-fluorescence test (IFT) for the presence of Leishmania-specific IgG antibodies. In the IFT IgG antibodies were detected after binding to L. major promastigotes via a DyLight 488-conjugated secondary antibody (Dianova, Hamburg, Germany) and analyzed with an Axiophot fluorescence microscope (Carl Zeiss, Göttingen, Germany). To confirm the results from IFT, a Leishmania Western Blot kit (BioRepair, Sinsheim, Germany) was used according to the manufacturer’s instructions; serum IgG was detected by rabbit anti-horse, rabbit anti-dog and goat anti-cat IgG conjugated to alkaline phosphatase (Dianova). As a reference the 14 and 16 kDa bands of anti-Leishmania IgG were used. In order to screen for the presence of viable Leishmania parasites, EDTA blood was diluted in 1:2 dilution steps with Leishmania culture medium [Schneider’s Drosophila insect cell medium C4200 (Genaxxon Bioscience) supplemented with 10 mM Hepes, 10% FCS, 2% human urine, 4% AAP solution containing 75 mg/ml penicillin G, 162.5 mg/ml streptomycin, 25 mM sodium pyruvate, 50 mM L-glutamine, 50 mM DMEM, 6.8 mM L-asparagine, 13.8 mM L-arginine] and incubated at 28 °C for 14 days until microscopic analysis.
Results
Focus regions for sand fly trapping in Bavaria
Seven focus regions were choosen for the survey of sand flies in Bavaria and allocated to the following administrative districts (Fig. 1): (1) Aschaffenburg/Main-Spessart; (2) Würzburg/Kitzingen; (3) Erlangen-Höchstadt/Forchheim; (4) Regensburg/Kehlheim; (5) Aichach-Friedberg/Landsberg am Lech/Weilheim-Schongau; (6) Passau; (7) Lindau am Bodensee/Oberallgäu. The rationale to focus on these regions was their predicted suitability for the establishment of the sand fly vector as obtained by regional distribution models based on present and future climate parameters (Fischer et al. 2010) (focus regions 1-7). Further criteria were reported cases of autochthonous leishmaniasis in man, dog and horse in the Bavarian administrative districts of Ostallgäu, Landsberg am Lech and Augsburg (Gothe 1991, Bogdan et al. 2001, Koehler et al. 2002) (focus region 5; Fig. 1); evidence for sand flies in the bordering countries Switzerland (Grimm et al. 1993) and Austria as well as in the German Federal State of Baden-Württemberg (Naucke et al. 2008, Naucke et al. 2011) (focus regions 1, 6-7; Fig. 1); and a relatively high present mean annual temperature of some regions compared to the rest of Bavaria (Naucke et al. 2008) (focus regions 1- 4).
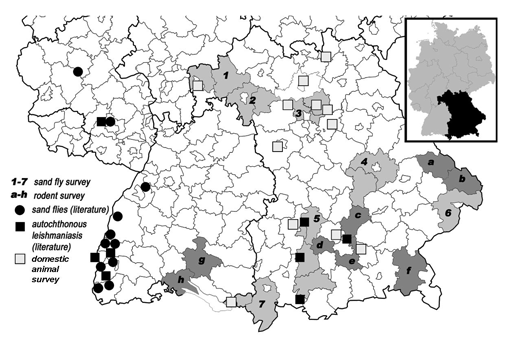
Fig. 1. Map of southern Germany with sand fly and rodent trapping sites. The insert shows a map of Germany with the State of Bavaria marked in black. CDC light traps for sand fly trapping were put up in seven focus areas in Bavaria (indicated by the numbers 1-7 and shaded in light gray). 1, Aschaffenburg/Main-Spessart; 2, Würzburg/Kitzingen; 3, Erlangen-Höchstadt/Forchheim; 4, Regensburg/Kehlheim; 5, Aichach-Friedberg/Landsberg am Lech/Weilheim-Schongau; 6, Passau; 7, Lindau am Bodensee/Oberallgäu. Rodents that were screened for Leishmania-infection were trapped in six administrative districts in Bavaria and two districts in Baden-Württemberg (indicated by the letters a-h and shaded in dark gray). a, Regen; b) Freyung-Grafenau; c) Freising; d) Fürstenfeldbruck; e) München; f) Traunstein; g) Sigmaringen; h) Konstanz. Locations of veterinary clinics that provided blood samples from domestic animals as well as sites of reported cases of autochthonous leishmaniasis in Bavaria and Baden-Württemberg, and some of the reported sites of sand flies in Baden-Württemberg are marked (Gothe 1991, Bogdan et al. 2001, Koehler et al. 2002, Naucke et al. 2008).
Prevalence of sand flies in Bavaria
In the study period 2009-2010 a total of 202 CDC light traps were put up in 155 different Bavarian towns (Table 1). Thereof, 187 traps were located inside the seven focus regions, 15 traps were mounted in other administrative districts (Ansbach, Ebersberg, Rosenheim, Weißenburg-Gunzenhausen). In 2009 only 58 traps in 40 towns were put up because of a cold and rainy summer season which was assumed to counteract sand fly activity since the predicted sand fly species need a monthly mean temperature of 18-20 °C in July (Aspöck et al. 2006). In 2010, 144 traps were mounted in 120 towns. Each trap contained an estimated number of 50-200 insects, with the majority being dipterans, including different mosquito species (Culex spp.) and numerous specimens of the Psychodidae family (Psychoda spp.), which includes the genus Phlebotomus. However, not a single sand fly was detected, even in the districts where sporadic autochthonous cases of leishmaniasis had been reported in the previous years (Gothe 1991, Bogdan et al. 2001, Koehler et al. 2002).
| Focus regiona | Administrative districts | Number of trapsb (number of towns) | ||
|---|---|---|---|---|
| in 2009 | in 2010 | TOTAL | ||
| 1 | Aschaffenburg/Main-Spessart | 0 (0) | 16 (12) | 16 (12) |
| 2 | Würzburg/Kitzingen | 0 (0) | 19 (10) | 19 (10) |
| 3 | Erlangen-Höchstadt/Forchheim | 23 (13) | 30 (26) | 53 (35d) |
| 4 | Regensburg/Kehlheim | 7 (4) | 26 (26) | 33 (29d) |
| 5 | Aichach-Friedberg/Landsberg am Lech/ Weilheim-Schongau | 8 (7) | 24 (20) | 32 (27) |
| 6 | Passau | 5 (4) | 24 (22) | 29 (26) |
| 7 | Lindau am Bodensee/Oberallgäu | 0 (0) | 5 (4) | 5 (4) |
| - | Othersc | 15 (12) | 0 (0) | 15 (12) |
| TOTAL | 58 (40) | 144 (120) | 202 (155d) | |
a Focus regions were defined on the basis of epidemiological and climate parameters.
b CDC light traps were mounted in 2009-2010 during June-August overnight in stables and old barns and analyzed for sand flies. No sand fly was trapped.
c Administrative districts: Ansbach, Ebersberg, Rosenheim, Weißenburg-Gunzenhausen
d Some towns were surveyed in both years.
Establishment of a protocol for the screening of Leishmania-DNA in wild rodents
Different methods for nucleic acid isolation were evaluated and compared for the yield and purity of total DNA extracted from tissue homogenates of wild rodents. DNA isolation using the trizol reagent showed a high yield and purity, but was inappropriate due to a certain degree of DNA degradation as revealed by gel electrophoresis. The DNA yield was poor using the QIAamp Viral RNA Mini Kit. Therefore, DNA from murine tissue homogenates was isolated using the AllPrep DNA/RNA Kit which showed both a sufficient high DNA yield and purity. For the detection of Leishmania DNA within the rodent DNA samples, four different PCR protocols were tested. Real-time RT-PCR of the Leishmania SSU rDNA as well as conventional PCRs using primers for either kinetoplast or 18S rDNA were cross-reactive for DNA from the related protozoan Trypanosoma grosi (four of twelve rodent samples were positive as identified by sequencing, data not shown). Therefore, all rodent DNA samples were screened using a conventional mini exon PCR which was specific for Leishmania DNA.
Prevalence of Leishmania-infection in rodents
A total of 300 rodents and 2 insectivores that had been captured between 2001 and 2008 in Bavaria and Baden-Württemberg (Fig. 1) were screened by mini exon PCR for the presence of Leishmania DNA in liver and ear skin samples. Six species of rodents were analyzed: 140 specimens of Bank voles (Myodes glareolus), 127 Yellow-necked mice (Apodemus flavicollis), 17 Wood mice (Apodemus sylvaticus), 14 Common voles (Microtus arvalis), 1 House mouse (Mus musculus), and 1 European water vole (Arvicola amphibius). In addition, two species of insectivores were screened: 1 Common shrew (Sorex araneus) and 1 Crowned shrew (Sorex coronatus). Infection of A. flavicollis with the rodent pathogen Trypanosoma grosi was detected by sequencing (data not shown). However, none of the tissue samples was positive for Leishmania (Table 2), even samples from districts that were close to the sites of autochthonous leishmaniasis in dog and horse that were reported previously (Gothe 1991, Koehler et al. 2002).
| Area | Administrative district | State | Number of rodents analyzeda | |||||
|---|---|---|---|---|---|---|---|---|
| A. fla-vicollis | A. syl-vaticus | Mi. ar-valis | M. gla-reolus | Othersc | TOTAL (Leishmania-positive)b | |||
| a | Regen | BY | 11 | 2 | 1 | 7 | 1 | 22 (0) |
| b | Freyung-Grafenau | BY | 2 | 2 | 0 | 14 | 0 | 18 (0) |
| c | Freising | BY | 0 | 0 | 1 | 7 | 0 | 8 (0) |
| d | Fürstenfeld-bruck | BY | 15 | 3 | 0 | 6 | 1 | 25 (0) |
| e | München | BY | 4 | 2 | 0 | 0 | 0 | 6 (0) |
| f | Traunstein | BY | 4 | 0 | 0 | 21 | 0 | 25 (0) |
| g | Sigmaringen | BW | 87 | 5 | 11 | 85 | 2 | 190 (0) |
| h | Konstanz | BW | 1 | 3 | 1 | 0 | 0 | 5 (0) |
| - | Othersd | BY | 3 | 0 | 0 | 0 | 0 | 3 (0) |
| TOTAL | 127 | 17 | 14 | 140 | 4 | 302 (0) | ||
a Rodents and insectivores were trapped in the years 2001-2008 in different regions of Bavaria (BY) and Baden-Württemberg (BW).
b Ear skin and liver samples from rodents and insectivores were analyzed by mini exon PCR for Leishmania infection.
c Rodents: House mouse (Mus musculus), European water vole (Arvicola amphibius); insectivores: Common shrew (Sorex araneus), Crowned shrew (Sorex coronatus)
d Administrative districts: Altötting, Erding
Prevalence of Leishmania infection in domestic animals
A systematic survey for the prevalence of autochthonous leishmaniasis in domestic animals in Bavaria was performed by screening serum and EDTA blood samples from horses, dogs and cats with negative travel history (i.e. no stay abroad, parents originate from Germany) in order to exclude imported leishmaniasis cases. Samples were provided by veterinarians from 12 administrative districts from throughout Bavaria (Fig. 1). In total 46 animals were screened (25 horses, 10 dogs, 11 cats). Only one horse showed clinical symptoms of leishmaniasis (nodular skin lesions, loss of hairs), all others were asymptomatic. However, all samples from both symptomatic and asymptomatic animals were negative for Leishmania-specific antibodies as revealed by Western Blot and IFT from serum samples. In addition, the culture from EDTA blood was negative (Table 3).
| Administrative district | Number of domestic animals analyzed | |||
|---|---|---|---|---|
| Horses | Dogs | Cats | TOTAL (Leishmania-positive)a | |
| Altötting | 1b | 0 | 0 | 2 (0) |
| Ansbach | 0 | 3 | 0 | 3 (0) |
| Augsburg | 0 | 1 | 1 | 2 (0) |
| Bamberg | 1 | 0 | 0 | 1 (0) |
| Dachau | 0 | 2 | 8 | 10 (0) |
| Ebersberg | 3 | 0 | 0 | 3 (0) |
| Forchheim | 0 | 3 | 0 | 3 (0) |
| Lichtenfels | 3 | 0 | 0 | 3 (0) |
| Lindau | 1 | 0 | 2 | 3 (0) |
| Miltenberg | 4 | 0 | 0 | 4 (0) |
| Neustadt/Aisch | 5 | 1 | 0 | 6 (0) |
| Nürnberger Land | 7 | 0 | 0 | 7 (0) |
| TOTAL | 25 | 10 | 11 | 46 (0) |
a Serum samples were screened by Western Blot and IFT. EDTA blood samples were screened by culture in Leishmania culture medium.
b Symptomatic animal
Discussion
The starting hypothesis of this study was that Leishmania vectors and Leishmania-infected wild and domestic reservoir hosts are present in Bavaria which was suggested by the evidence for previous autochthonous cases of leishmaniasis in this area. However, no sand flies and no infected rodents or domestic animals were detected. The obvious absence of sand flies in the surveyed years 2009 and 2010 is on the one hand surprising because without a vector autochthonous Leishmania transmission cannot occur; on the other hand it is in accordance with distribution models by the group of Carl Beierkuhnlein (Fischer et al. 2010) which indicate that the current temperature conditions within Bavaria are unsuitable for the occurrence of sand flies. However, already in the next few years, as modelled for the period 2011-2040, the temperature requirements will be partially fulfilled in certain Bavarian regions, most of which have been surveyed in the present study. Very recently, a similar approach gave the first evidence for sand flies in Austria (Naucke et al. 2011) in a non-endemic region that was previously defined by climatic models to be suitable for sand fly occurrence (Aspöck et al. 2007).
If Bavaria is presently not populated by sand flies, how should the recorded sporadic occurrence of autochthonous leishmaniasis cases in Bavaria be explained? These cases were recorded in 1991 (Gothe 1991), 1997 (Bogdan et al. 2001) and 2000 (Koehler et al. 2002). From our field study in the years 2009 and 2010, we cannot exclude a periodically presence of sand flies in Bavaria in earlier years. Of note, the climatic conditions in Germany were especially favorable at the end of the 20th century, that is when all the autochthonous cases occurred: in the whole period of 1901 until 2008, the peak in the rise of the annual mean temperature was between 1981 and 2000, with a dramatic 2.3 °C increase in the cold season (Jonas et al. 2005, DWD 2009). The uncommon mild winters could have been favorable for sand fly over-wintering.
In a second approach, rodents were analyzed for the prevalence of Leishmania infection. Rodents have been identified as wild reservoir hosts for Leishmania parasites in disease-endemic countries (Rassi et al. 2006, Pourmohammadi et al. 2008, Papadogiannakis et al. 2010). Thus, Leishmania positive rodents might serve as an indirect indicator for the presence of Leishmania-transmitting sand flies. In total, ear skin and liver samples from 300 rodents and 2 insectivores out of eight different administrative districts in Bavaria and Baden-Württemberg were analyzed for the presence of Leishmania-DNA by the use of a highly sensitive Leishmania-specific PCR, but none of the samples was positively tested for Leishmania-DNA. Thus, the prevalence of Leishmania in rodents of this area can be considered as nonexistent or at least very low. However, other species of rodents and small mammals that were not surveyed in this study might nevertheless serve as natural reservoir hosts. L. infantum infection was described for the Brown rat (Rattus norvegicus) in Greece (Papadogiannakis et al. 2010, Psaroulaki et al. 2010), for the Black rat (Rattus rattus) in Spain and Italy (Gradoni et al. 1983, Martin-Sanchez et al. 2004), and in the Mediterranean island of Mallorca 39% of all tested pine martens (weasel family) were positive for L. infantum (Millan et al. 2011). All three mammal species are also abundant in Germany and might thus be useful disease sentinels for the identification of new endemic foci (Psaroulaki et al. 2010).
Since most of the previous autochthonous cases of leishmaniasis in Bavaria in the years 1991 to 2000 were diagnosed in domestic animals (two dogs, one horse) (Gothe 1991, Koehler et al. 2002), a spread of leishmaniasis should be detected most easily in an increased number of autochthonous infections of domestic reservoir animals without travel history. However, none of the screened dogs, cats and horses from the years 2009 and 2010 was positive for Leishmania infection. This demonstrates that in contrast to the high number of imported cases of canine leishmaniasis (Menn et al. 2010) autochthonous cases are still a very sporadic event.
Taken together, Bavaria is presently not an established area for the occurrence of sand flies and for autochthonous leishmaniasis of wild rodents and domestic animals. The risk for autochthonous leishmaniasis in Bavaria can therefore be considered as low. However, since temperature requirements are the most important habitat factor for sand flies (Cross and Hyams 1996, Aspock et al. 2008, Fischer et al. 2010) and steadily rising temperatures due to climate change are a fact, a spread of the phlebotomine vector to Germany is very likely and strongly supported by temperature-derived vector distribution models also for the Federal State of Bavaria (Fischer et al. 2010). In addition, the density of rodents – a potential parasite reservoir – is expected to increase in southern Germany as a consequence of climate change (Piechotowski et al. 2008). Therefore, we recommend future surveys on the presence of sand flies in Bavaria and surveys on the establishment of rodents as reservoir for leishmaniasis besides domestic animals.
References
Aspock, H, Gerersdorfer, T, Formayer, H, Walochnik, J. Sandflies and sandfly-borne infections of humans in Central Europe in the light of climate change. Wien Klin Wochenschr 2008; 120:24-29.
Aspöck, H, Walochnik, J, Gerersdorfer, T, Formayer, H. Risikoprofil für das autochthone Auftreten von Leishmaniosen in Österreich. StarClim2006.B. 2006. Available at: http://www.austroclim.at/startclim
Bogdan, C, Schonian, G, Banuls, AL, Hide, M et al. Visceral leishmaniasis in a German child who had never entered a known endemic area: case report and review of the literature. Clin Infect Dis 2001; 32:302-306.
Cross, ER, Hyams, KC. The potential effect of global warming on the geographic and seasonal distribution of Phlebotomus papatasi in southwest Asia. Environ Health Perspect 1996; 104:724-727.
DWD. Klimawandel im Detail – Zahlen und Fakten zum Klima in Deutschland. Deutscher Wetterdienst. 2009. Available at: http://www.dwd.de/bvbw/generator/DWDWWW/Content/Presse/Pressekonferenzen/2008/PK-15-04-2008/20080415__Z_2BF__PDF__download,templateId=raw,property=publicationFile.pdf/20080415_Z+F_ PDF_download.pdf
Englund, L, Pringle, J. New diseases and increased risk of diseases in companion animals and horses due to transport. Acta Vet Scand Suppl 2003; 100:19-25.
Fischer, D, Thomas, SM, Beierkuhnlein, C. Temperature-derived potential for the establishment of phlebotomine sandflies and visceral leishmaniasis in Germany. Geospat Health 2010; 5:59-69.
Gothe, R. Leishmaniosen des Hundes in Deutschland: Erregerfauna und -biologie, Epidemiologie, Klinik, Pathogenese, Diagnose, Therapie und Prophylaxe. Kleintierpraxis 1991; 36:69-84
Gradoni, L, Pozio, E, Gramiccia, M, Maroli, M et al. Leishmaniasis in Tuscany (Italy): VII. Studies on the role of the black rat, Rattus rattus, in the epidemiology of visceral leishmaniasis. Trans R Soc Trop Med Hyg 1983; 77:427-431.
Grimm, F, Gessler, M, Jenni, L. Aspects of sandfly biology in southern Switzerland. Med Vet Entomol 1993; 7:170-176.
Jonas, M, Staeger, T, Schönwiese, CD. Berechnung der Wahrscheinlichkeiten für das Eintreten von Extremereignissen durch Klimaänderungen - Schwerpunkt Deutschland. Climate Change - Forschungsbericht 07/05. Umweltbundesamt. 2005. Available at: http://www.umweltbundesamt.de/uba-info-medien/dateien/2946.htm
Killick-Kendrick, R. Phlebotomine vectors of the leishmaniases: a review. Med Vet Entomol 1990; 4:1-24.
Koehler, K, Stechele, M, Hetzel, U, Domingo, M et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109:9-17.
Lozán, JL, Garms, R, Naucke, TJ. Die Leishmaniose - eine potenzielle Gefahr in Mitteleuropa. In: Lozán, JL, Graßl, H, Jendritzky, G, Karbe, L, Reise, K, eds. Warnsignal Klima: Gesundheitsrisiken. Hamburg, 2008: p 211-214.
Maia, C, Gomes, J, Cristovao, J, Nunes, M et al. Feline Leishmania infection in a canine leishmaniasis endemic region, Portugal. Vet Parasitol 2010; 174:336-340.
Martin-Sanchez, J, Gramiccia, M, Di Muccio, T, Ludovisi, A et al. Isoenzymatic polymorphism of Leishmania infantum in southern Spain. Trans R Soc Trop Med Hyg 2004; 98:228-232.
Menn, B, Lorentz, S, Naucke, TJ. Imported and travelling dogs as carriers of canine vector-borne pathogens in Germany. Parasit Vectors 2010; 3:34.
Millan, J, Zanet, S, Gomis, M, Trisciuoglio, A et al. An investigation into alternative reservoirs of canine leishmaniasis on the endemic island of mallorca (Spain). Transbound Emerg Dis 2011; 58:352-357.
Naucke, TJ, Lorentz, S, Rauchenwald, F, Aspock, H. Phlebotomus (Transphlebotomus) mascittii Grassi, 1908, in Carinthia: first record of the occurrence of sandflies in Austria (Diptera: Psychodidae: Phlebotominae). Parasitol Res 2011; 109:1161-1164.
Naucke, TJ, Menn, B, Massberg, D, Lorentz, S. Sandflies and leishmaniasis in Germany. Parasitol Res 2008; 103 Suppl 1:S65-68.
Naucke, TJ, Pesson, B. Presence of Phlebotomus (Transphlebotomus) mascittii Grassi, 1908 (Diptera : Psychodidae) in Germany. Parasitol Res 2000; 86:335-336.
Papadogiannakis, E, Spanakos, G, Kontos, V, Menounos, PG et al. Molecular detection of Leishmania infantum in wild rodents (Rattus norvegicus) in Greece. Zoonoses Public Health 2010; 57:e23-25.
Piechotowski, I, Brockmann, SO, Schwarz, C, Winter, CH et al. Emergence of hantavirus in South Germany: rodents, climate and human infections. Parasitol Res 2008; 103 Suppl 1:S131-137.
Pourmohammadi, B, Motazedian, MH, Kalantari, M. Rodent infection with Leishmania in a new focus of human cutaneous leishmaniasis, in northern Iran. Ann Trop Med Parasitol 2008; 102:127-133.
Psaroulaki, A, Antoniou, M, Toumazos, P, Mazeris, A et al. Rats as indicators of the presence and dispersal of six zoonotic microbial agents in Cyprus, an island ecosystem: a seroepidemiological study. Trans R Soc Trop Med Hyg 2010; 104:733-739.
Quinnell, RJ, Courtenay, O. Transmission, reservoir hosts and control of zoonotic visceral leishmaniasis. Parasitology 2009; 136:1915-1934.
Rassi, Y, Javadian, E, Amin, M, Rafizadeh, S et al. Meriones libycus is the main reservoir of zoonotic cutaneous leishmaniasis in south Islamic Republic of Iran. East Mediterr Health J 2006; 12:474-477.
Ready, PD. Leishmaniasis emergence and climate change. Rev Sci Tech 2008; 27:399-412.
Rodgers, MR, Popper, SJ, Wirth, DF. Amplification of kinetoplast DNA as a tool in the detection and diagnosis of Leishmania. Exp Parasitol 1990; 71:267–275.
Schex, S, Dobler, G, Riehm, J, Müller, J et al. Rickettsia spp. in wild small mammals in Lower Bavaria, South-Eastern Germany. Vector Borne Zoonotic Dis 2011; 11:493-502.
Schulz, A, Mellenthin, K, Schönian, G, Fleischer, B et al. Detection, Differentiation, and Quantitation of Pathogenic Leishmania Organisms by a Fluorescence Resonance Energy Transfer-Based Real-Time PCR Assay. J Clin Microbiol 2003; 41:1529–1535.
Weitzel, T, Muhlberger, N, Jelinek, T, Schunk, M et al. Imported leishmaniasis in Germany 2001-2004: data of the SIMPID surveillance network. Eur J Clin Microbiol Infect Dis 2005; 24:471-476.
Wortmann, G, Sweeney, C, Houng, HS, Aronson, N et al. Rapid diagnosis of leishmaniasis by fluorogenic polymerase chain reaction. Am J Trop Med Hyg 2001; 65:583-587.
Publications during the funding period
Bogdan C. Mechanisms and consequences of persistence of intracellular pathogens: leishmaniasis as an example. Cell Microbiol. 2008, 10(6):1221-34.
Liese J, Schleicher U and Bogdan C. The innate immune response against Leishmania parasites. Immunobiology 2008, 213 (3-4): 377-387.
Boecken G, Weitzel T, Sunderkötter C, Fischer M, von Stebut-Borschitz E, Bogdan
C, Pietras M, Anders G, Harms-Zwingenberger G, Burchard G, Bialek R, Lippert U,
Grobusch M, Erkens K, Fleischer B, Löbermann M, Schunk M, Sterzik B. Diagnosis
and therapy of cutaneous and mucocutaneous Leishmaniasis in Germany . J Dtsch
Dermatol Ges. 2009 Oct;7 Suppl 7:s1-38.
Boecken G, Sunderkötter C, Bogdan C, Weitzel T, Fischer M, Müller A, Löbermann M, Anders G, von Stebut E, Schunk M, Burchard G, Grobusch M, Bialek R, Harms-Zwingenberger G, Fleischer B, Pietras M, Faulde M, Erkens K. S1 Guideline: Diagnosis and therapy of cutaneous and mucocutaneous leishmaniasis in Germany. J. Dtsch Dermatol Ges. 2011 (in press)
Bogdan C. Leishmaniasis in rheumatology, hematology and oncology: epidemicological, immunological and clinical aspects and caveats. Annals Rheumatic Diseases 2011 (in press).
Amann K, Bogdan C, Harrer T and Rech J. Renal leishmaniasis as unusual cause of nephrotic syndrome in a HIV patient. J Am Soc Nephrology 2011 (in press).
Simone Haeberlein, Karin Knoll, Sandra Essbauer, Dominik Fischer, Stephanie Margarete Thomas, Carl Beierkühnlein, Ulrike Schleicher, Christian Bogdan. Autochthonous leishmaniasis in Germany: survey of the prevalence of sand flies and infected rodents. Manuscript in preparation (2011).
Presentations on conferences and workshops
Christian Bogdan "Vernachlässigte Infektionskrankheiten: Leishmaniose", Hauptvortrag 9. Kongress für Infektiologie und Tropenmedizin, Innsbruck, 28. Februar 2008
Christian Bogdan "Erderwärmung: Wegbereiter für die Ausbreitung (tropischer) bakterieller und parasitärer Infektionskrankheiten in Deutschland?", Hauptvortrag, Klinisch-Infektiologische Fortbildungsreihe des Klinikums Erlangen für Ärzte in Mittelfranken, Erlangen, 28. Mai 2008
Christian Bogdan "Standards der Diagnostik und Therapie der Leishmaniosen", Hauptvortrag, 28. Bayerische Chemotherapie-Expertengespräche, Neufarn, 4. Juli 2008
Simone Häberlein "Leishmaniose in Bayern? Untersuchungen zur Vektorprävalenz und zur Existenz tierischer Reservoirs", Klimawoche Bayern, München, 17. Juli 2009
Christian Bogdan „Epidemiology, Immunology and Chemotherapy of leishmaniasis“,
Hauptvortrag, 61. Jahrestagung der Deutschen Gesellschaft für Hygiene und
Mikrobiologie, Göttingen, 21. September 2009
Simone Häberlein "Orientbeulen und andere Leishmaniosen: Mitbringsel aus dem
Ausland", Lange Nacht der Wissenschaft, 24. Oktober 2009, Universität Erlangen-
Nürnberg
Christian Bogdan "Autochthone Leishmaniose in Bayern: Untersuchungen zur
Vektorprävalenz und zur Existenz tierischer Reservoirs", VICCI Workshop
München, 13. November 2009
Christian Bogdan "Chronische Hautinfektionen", Hauptvortrag, Klinisch-
Infektiologische Fortbildungsreihe des Klinikums Erlangen für Ärzte in Mittelfranken,
24. März 2010
Simone Häberlein "Autochthone Leishmaniose in Bayern: Untersuchungen zur
Vektorprävalenz und zur Existenz tierischer Reservoirs", VICCI Klausur-Workshop,
St. Oswald, 25. März 2010
Christian Bogdan "Epidemiologie, Klinik, Diagnostik und Therapie der kutanen
Leishmaniose, Fortbildungsvortrag für Hautärzte der Universitätsklinik Erlangen, 5.
Mai 2010
Christian Bogdan „In Bayern und Deutschland erworbene Leishmaniose – gibt es das?“, Klimawoche Bayern, Erlangen, 27. Juli 2010
Christian Bogdan "Autochthone Leishmaniose in Bayern: Untersuchungen zur
Vektorprävalenz und zur Existenz tierischer Reservoirs", VICCI Klausur-Workshop,
St. Oswald, 17. Mai 2011
Christian Bogdan „Krank durch Sandmücken: Zur Epidemiologie, Klinik und Diagnostik der Leishmaniose“, Klimawoche Bayern, München, 8. Juni 2011
Simone Haeberlein, Karin Knoll, Sandra Essbauer, Ulrike Schleicher, Christian Bogdan. Autochthonous leishmaniasis in Germany: survey of the prevalence of sand flies and infected rodents in Bavaria. Posterbeitrag 63. Jahrestagung der Deutschen Gesellschaft für Hygiene und Mikrobiologie, Essen, 26. September 2011
Mehr zu diesem Thema
Allgemeine Informationen zum Thema
- (VICCI) Forschungsverbund: Ziel, Hintergrund, Ansprechpartner
Projektergebnisse
Weitere (VICCI)-Projekte
- (VICCI) Projekt 1: Prospektive Studie zur Entwicklung von Borrelia burgdorferi s.l. Spezies in Ixodes ricinus in Bayern
- (VICCI) Projekt 2: Modellierung des Vorkommens zeckenübertragener Krankheitserreger in bayerischen Naherholungsgebieten
- (VICCI) Projekt 3: Studie und epidemiologisches Computermodelling von zeckenübertragenen Erkrankungen in Bayern
- (VICCI) Projekt 4: Risikoabschätzung zeckenübertragener bakterieller Infektionskrankheiten in urbanen Parkanlagen Bayerns
- (VICCI) Projekt 5: Studie zum Vorkommen Nagetier-Übertragener Zoonosen entlang eines Klimagradienten im Nationalpark Bayerischer Wald
- (VICCI) Projekt 7: Biogeographische Analyse gesundheitsrelevanter Arten und Prognose ihres Ausbreitungspotenzials in Bayern unter veränderten künftigen Klimabedingungen
- (VICCI) Projekt 8.1: Querschnittsprojekt Datenzentrum
- (VICCI) Projekt 8.2: Bevölkerungsbezogene epidemiologische Risikoabschätzung
- (VICCI) Projekt 8.3: Verbundorganisation


